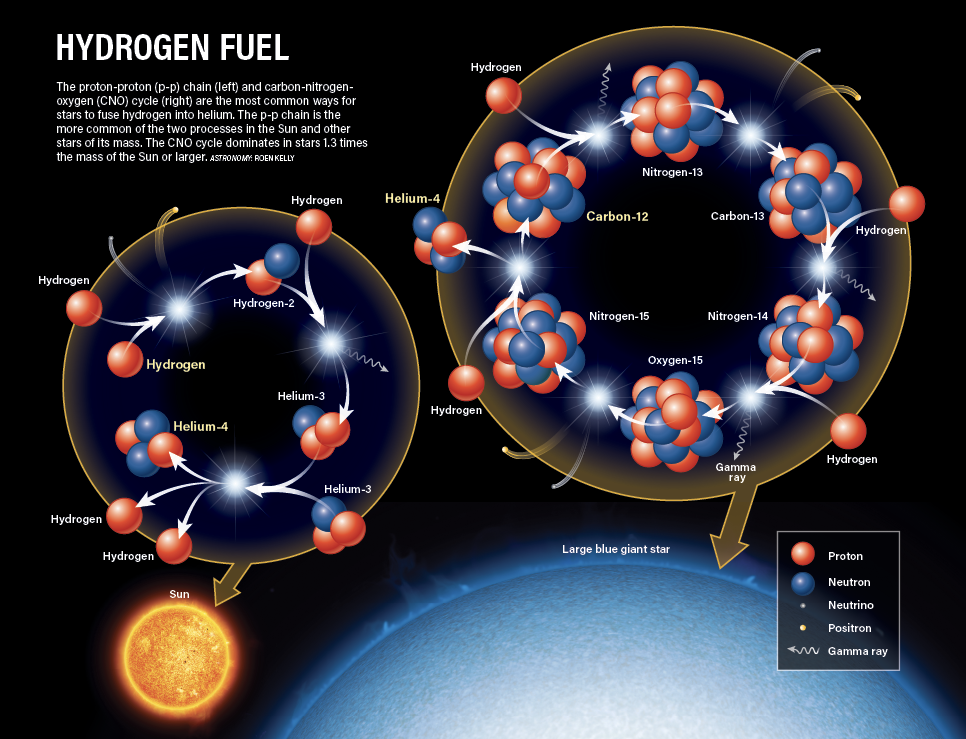
Other than hydrogen and helium, oxygen is the most common element in the universe (followed closely by carbon). Hydrogen and helium condensed from light itself during the Big Bang, but everything else is made in the core of stars. Three helium atoms make carbon, and four helium atoms make oxygen.
The Earth’s crust and mantle are made of rocks, which are made of minerals. Each mineral is a different chemical compound whose atoms are arranged in a regular 3D crystal “lattice.” To a good approximation, rocks are composed of oxygen atoms that are crammed as close together as possible, and all the other elements occupy just the little void spaces between the oxygen spheres!
When the Earth was young, all of the oxygen was chemically bound up like this. The only oxygen in the early atmosphere was bound up too, mostly as CO2 and H2O. The early atmosphere was what chemists call a “reducing” environment. There was virtually no free oxygen in the air for the first couple of billion years of Earth’s history, but there were other gases that couldn’t exist today: ammonia, hydrogen, methane, hydrogen sulfide. It would have been toxic, but there were no living things at the surface to be harmed.
Not until at least a billion years after Earth’s formation did the first photosynthetic microbes in the oceans begin to free oxygen from its chemical bonds. The biggest chemical change in geologic history had begun — the oxidation of the oceans, atmosphere, and even the Earth’s crust!